 In doing so, Lavoisier championed the idea of conservation of mass during transformations (Figure 3). However, during the second half of the 18th century, Lavoisier performed many quantitative experiments and observed that while substances changed form during a chemical reaction, the mass of the system – or a measure of the total amount of “stuff” present – did not change. Most of the theories that existed to explain the way that substances changed relied upon Greek philosophy, and there was precious little experimental detail attached to the alchemist’s tinkering. At the time, chemistry still couldn’t be described as being a true, quantitative science. Lavoisier: Law of Mass ConservationĪntoine Lavoisier was a French nobleman in the 1700s who began to experiment with different chemical reactions. However, there are some important moments in history that have helped to make sense of it. Despite their misguided approach, many early alchemists performed foundational chemical experiments - transforming one substance into another, and so it is difficult to point to a specific date or event as the birth of the idea of an ordered, quantifiable chemical reaction. Figure 2: Aristotle believed that everything in the world was composed of four fundamental substances - air, earth, fire, and water.Īs such, they proposed, and spent generations trying to prove, that less expensive metals like copper and mercury could be turned into gold. Alchemists based their approach on Aristotle’s ideas that everything in the world was composed of four fundamental substances - air, earth, fire, and water (Figure 2). Simple stones, such as those that contained sulfur, seemed to magically burn and otherwise unimpressive minerals were transformed, like the ore cinnabar becoming an enchanting silvery liquid metal mercury when heated. Historical contextīeginning in the early Middle Ages, European and Persian philosophers became fascinated with the way that some substances seemed to “transmute” (or transform) into others. What were these transformations and how were they controlled? These questions could only be answered when the transition from alchemy to chemistry as a quantitative and experimental science took place. Processes like fermentation, in which sugars are chemically converted into alcohol, have been known for centuries however, the chemical basis of the reaction was not understood. 
Though chemical reactions have been occurring on Earth since the beginning of time, it wasn’t until the 18th century that the early chemists started to understand them. 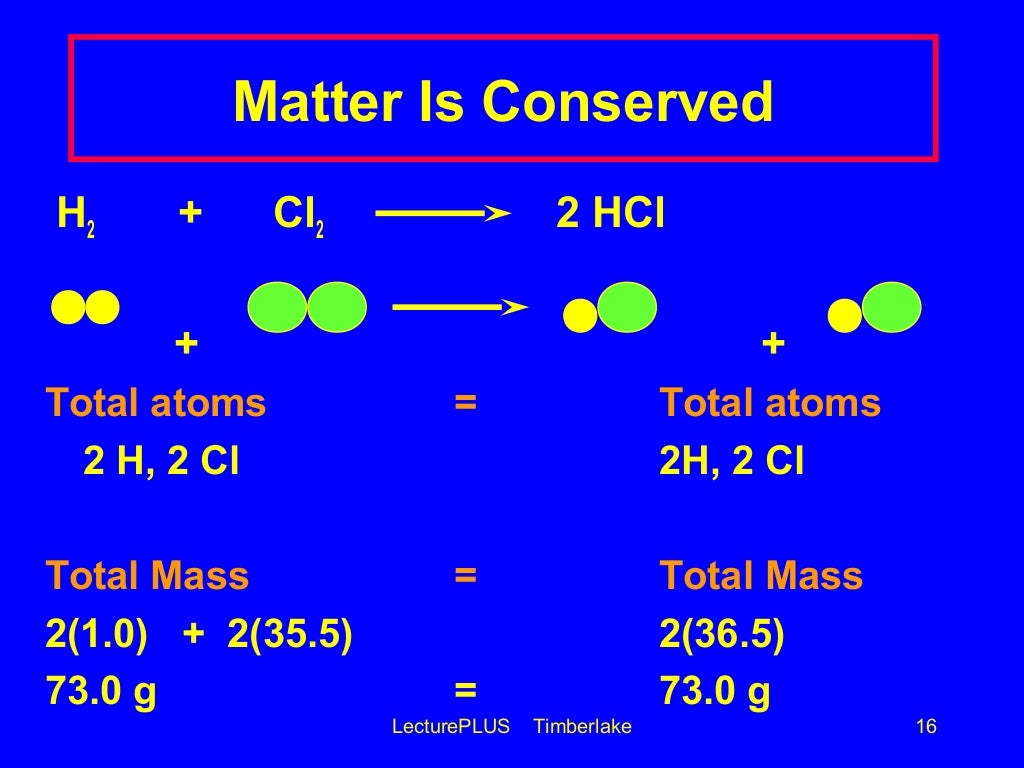
Figure 1: A controlled fire in Alberta, Canada, set to create a barrier for future wildfires. In nature, chemical reactions can be much less controlled than you’ll find in the lab, sometimes far messier, and they generally occur whether you want them to or not! Whether it be a fire raging across a forest (Figure 1), the slow process of iron rusting in the presence of oxygen and water over a period of years, or the delicate way in which fruit ripens on a tree, the process of converting one set of chemical substances (the reactants) to another set of substances (the products) is one known as a chemical reaction. In fact, the colossal number of transformations make for a dizzying, almost incomprehensible array of new substances and energy changes that take place in our world every second of every day. While we sometimes associate chemical reactions with the sterile environment of the test tube and the laboratory - nothing could be further from the truth. For the previous version, see this page.)Ĭhemical reactions happen absolutely everywhere. (This is an updated version of the Chemical Reactions module. 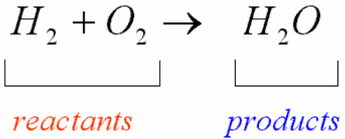
Understanding Scientific Journals and Articles. 
Using Graphs and Visual Data in Science.Scientists and the Scientific Community.Scientific Notation and Order of Magnitude.The Case of the Ivory-billed Woodpecker.Santiago Ramón y Cajal and Camillo Golgi.Factors that Control Earth's Temperature.Plates, Plate Boundaries, and Driving Forces.Solutions, Solubility, and Colligative Properties.Y-Chromsome and Mitochondrial DNA Haplotypes.Absorption, Distribution, and Storage of Chemicals. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
