Should a doctor or patient need to find the implant’s unique device identifier, an x-ray can read the notches. The technology is currently being used by Nvision, a Fountainhead portfolio company.įor spine devices, the company is “notching” or 3D barcoding tantalum markers. Fountainhead Investment Partners and its incubator Watershed Idea Foundry highlighted structural encoding technology that it plans to license to device companies as an alternative to sterile packaging or direct part marking. 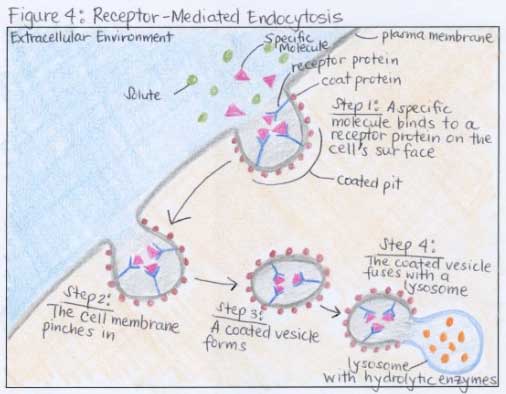
Unique Device Identification (UDI) remains a critical pain point for many device companies. For example, we can grow collagen on the surface of this plastic by loading it with copper the collagen cells will attract and proliferate on the surface.” “Introducing other ions in this platform will be the future of tissue regeneration. “We can also load strontium, magnesium and zinc into the same matrix and control how much is eluted,” Mr. hip and knee segments, where infection prevention is highly prevalent. CleanFuse material, an antimicrobial PEEK, has CE Mark approval, and the company plans to first launch it in U.S. In 1H18, the company plans to seek a strategic partner for ZFUSE.ĭiFusion’s patented process lends itself to development of bony ingrowth, antimicrobial and tissue regeneration polymers. Johns.ĭiFusion Technologies, founded in 2010, describes itself as an advanced biomaterials manufacturer and not a device company. No other company can make that claim, says Mr. 
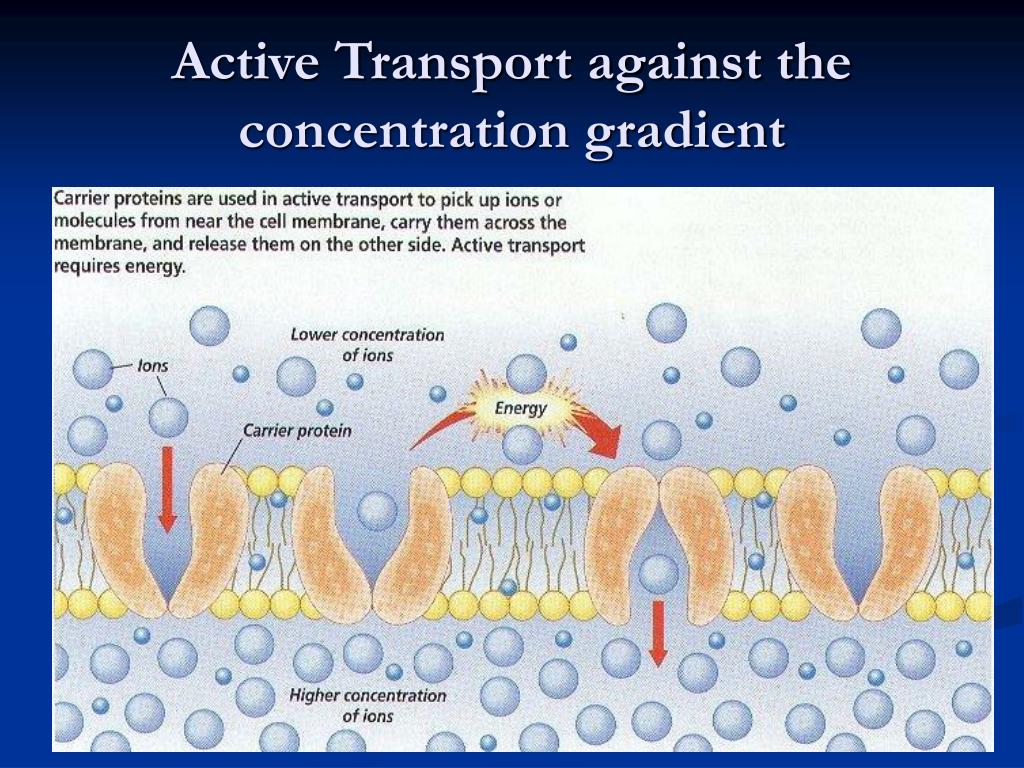
They expect to submit the data to FDA in January 2018 as part of a 510(k) filing that, once cleared, would give the company osteoconductive and biological fixation claims for their material. 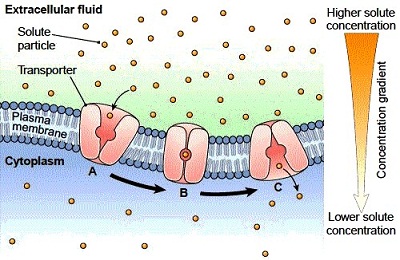
As spine companies seek alternatives to traditional PEEK, DiFusion President and CEO Derrick Johns says that the company merged the best qualities of titanium and PEEK to create ZFUSE.ĭiFusion Technologies recently invested $500,000 in a sheep study. We chose these five because they speak to the aforementioned technology trends and give insight into future product launches.ĭiFusion Technologies added zeolite molecules to Solvay’s Zeniva ZA-500 PEEK to change the material to a negatively-charged substance that cultivates osteoconduction while preserving the polymer’s visualization, modulus and strength benefits. Here’s a recap of just five conversations that we had on the exhibit hall floor. We know that hundreds of companies- thousands of people-were on hand at the 2017 event. While larger spine companies are already pursuing these technologies, we noticed that mid-sized down to startup players are prioritizing some or all in their product development pipelines. Transcription and Translation in ProkaryotesĪll molecules in a solution above the absolute zero temperature (-273.15☌) will be moving randomly.We met with surgeons, device companies and suppliers at the North American Spine Society (NASS) Annual Meeting and walked away with the impression that OEMs seek a broad portfolio that requires different manufacturing processes and products, other than hardware alone, to drive their revenue during a time of slow procedural volume.įor example, the topics that resonated in exhibit hall conversations and surgeon presentations were advancements in additive manufacturing or 3D printing, replacement or coating of traditional PEEK and the adoption of robotics and navigation.Changes in Signal Transduction Pathways. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
